|
CASE REPORT |
https://doi.org/10.5005/jp-journals-10080-1567
|
Efficacy of Temporary Intentional Leg Shortening and Deformation for Treatment of Massive Bone and Soft Tissue Defects in Three Patients
1–4Department of Orthopaedic Surgery, Graduate School of Medical Sciences, Kanazawa University, Kanazawa, Ishikawa, Japan
Corresponding Author: Hidenori Matsubara, Department of Orthopaedic Surgery, Graduate School of Medical Sciences, Kanazawa University, Kanazawa, Ishikawa, Japan, Phone: +81 762652374, e-mail: ortho331@yahoo.co.jp
How to cite this article: Ugaji S, Matsubara H, Aikawa T, et al. Efficacy of Temporary Intentional Leg Shortening and Deformation for Treatment of Massive Bone and Soft Tissue Defects in Three Patients. Strategies Trauma Limb Reconstr 2022;17(3):195–201.
Source of support: Nil
Conflict of interest: None
Received on: 02 September 2021; Accepted on: 22 August 2022; Published on: 30 December 2022
ABSTRACT
Aim: This study describes the intentional deformation and shortening of a limb using external fixation (EF) in three patients with post-traumatic injury tibial defects and to assess the outcomes.
Case description: Three patients with infected non-unions and massive bone and soft tissue defects who were treated with temporary intentional leg shortening and deformation using a Taylor Spatial Frame (TSF) EF were retrospectively reviewed. The alignment was restored by gradual deformity correction and lengthening after a 2-week interval. No additional surgical intervention was required for soft tissue reconstruction after primary skin closure. Skin closure and good bone alignment were achieved in all patients. The functional outcomes and bone outcomes were evaluated for all cases according to the Paley criteria modified by the Association for the Study and Application of Methods of Ilizarov. All patients showed excellent bone outcomes. Two patients achieved excellent functional outcomes and one had a good outcome. The patient with a good outcome was unable to descend the stairs comfortably.
Conclusion: This technique is suitable for treating massive bone and soft tissue defects and should be considered as a treatment option.
Keywords: Bone defect, External fixator, Infection, Taylor Spatial Frame, Tibia.
BACKGROUND
Massive bone and soft tissue defects are difficult to treat1,2 because both the defects require transplantation from other sites along with bone grafts; excellent surgical skills are necessary to perform these techniques. Treatment options include flaps,3 vascularised fibula grafts,2 and the Masquelet technique.4–7 However, many hospitals do not have orthoplastic teams to perform flap or microsurgery procedures. Free vascularised transfer of tissue involves a donor site morbidity and, despite this sacrifice, there are instances of flap failure. The Masquelet technique requires an intact soft tissue envelope and can only treat bone defects; this technique also requires a large amount of bone graft. This study aims to report the technique of temporary intentional leg shortening and deformation, which has been reported by Nho et al.8 in the English literature, and to assess the outcomes. We describe the advantages and disadvantages of this method.
CASE DESCRIPTION
The treatment method involves a temporary intentional leg shortening and deformation using an EF. This treatment can be used for a wide range of bone and soft tissue defects and, in these instances, uses a hexapod EF frame, the TSF. Devitalised bone and tissue are removed before the EF is applied; the limb is then manipulated, creating both a deformity and shortening, in order to approximate the ends of the wound and allow for primary closure (Fig. 1). At one week after this closure, a deformity correction protocol is initiated.

Figs 1A to E: Schema of intentional deformation and shortening. (A) Unnecessary bones and tissues are removed when covering the soft tissue; (B) External fixation is applied; (C) The deformation and shortening are made temporarily so as to cover the soft tissue; (D) Correcting deformity and lengthening; (E) Lengthening at the other side
Three patients with post-traumatic injury defects of the tibia were treated by intentional deformation and shortening using the TSF in this study. Two of the three patients were also treated with bone transport. Post-tibial assessment used the Paley criteria modified by Application of Methods of Ilizarov.9,10 The patients’ data are summarised in Table 1.
| Characteristics | Patient 1 | Patient 2 | Patient 3 |
|---|---|---|---|
| Age | |||
| Gustilo–Anderson classification | 3C | 3B | 3B |
| Bone loss (cm) | 4.5 | 5 | 7 |
| Soft tissue loss (cm) | 7 | 10 | 10 |
| EF period (day) | 84 | 110 | 364 |
| EF index (day/cm) | 15 | 22 | 52 |
| Follow-up (months) | 15 | 48 | 24 |
| Operation times | 2 | 3 | 3 |
| Bone result | Excellent | Excellent | Excellent |
| Functional result | Good | Excellent | Excellent |
Case 1
A 48-year-old male was injured by a concrete block that fell on his left lower leg. He sustained a Gustilo type 3C open fracture11,12 of the proximal tibia (Fig. 2A). The posterior tibial artery, peroneal artery, and anterior tibial artery were ruptured. An anastomosis of the posterior tibial and peroneal arteries was performed, and the tibia was shortened by 3 cm. A uniplanar EF was applied (Fig. 2B). Four months later, he was referred to our institution for further treatment of the 3-cm tibial shortening, an infected non-union and a residual soft tissue defect. Methicillin-sensitive Staphylococcus aureus was detected from the open wound and over-granulation had formed (Fig. 3A). The antibiotic cefazolin was administered intravenously for the methicillin-susceptible S. aureus. Debridement of the granulation tissue was performed first. This resulted in an anterior soft tissue defect measuring 7 cm by 2 cm. The non-union was exposed and resected. We then confirmed that the edges of the wound could be approximated by acute deformation of the limb (Fig. 3B). Three pre-constructed rings of TSF were applied; the anterior wound with exposed bone was closed by deforming the leg, creating a 25° apex–posterior angulation. An osteotomy at the distal part of the tibia was performed for lengthening to compensate for the limb shortening (Fig. 2C). Lengthening was commenced at the distal osteotomy (0.5 mm/day) 2 weeks postoperatively. The limb was kept in the deformed position for 3 weeks. A correction was commenced thereafter and took 6 weeks to complete (Fig. 2D) but the eventual 45-mm lengthening took 3 months to achieve equal leg lengths. The wound was healed completely at this time. The final procedure was to substitute the EF for plate fixation (Fig. 2E). Fifty months after the first operation (Fig. 2F), the patient was able to walk without pain (Fig. 3C). Using the outcome measures described earlier (modified criteria of Paley et al.), the bone result was excellent at 2 years after the procedure whereas the functional result was good.
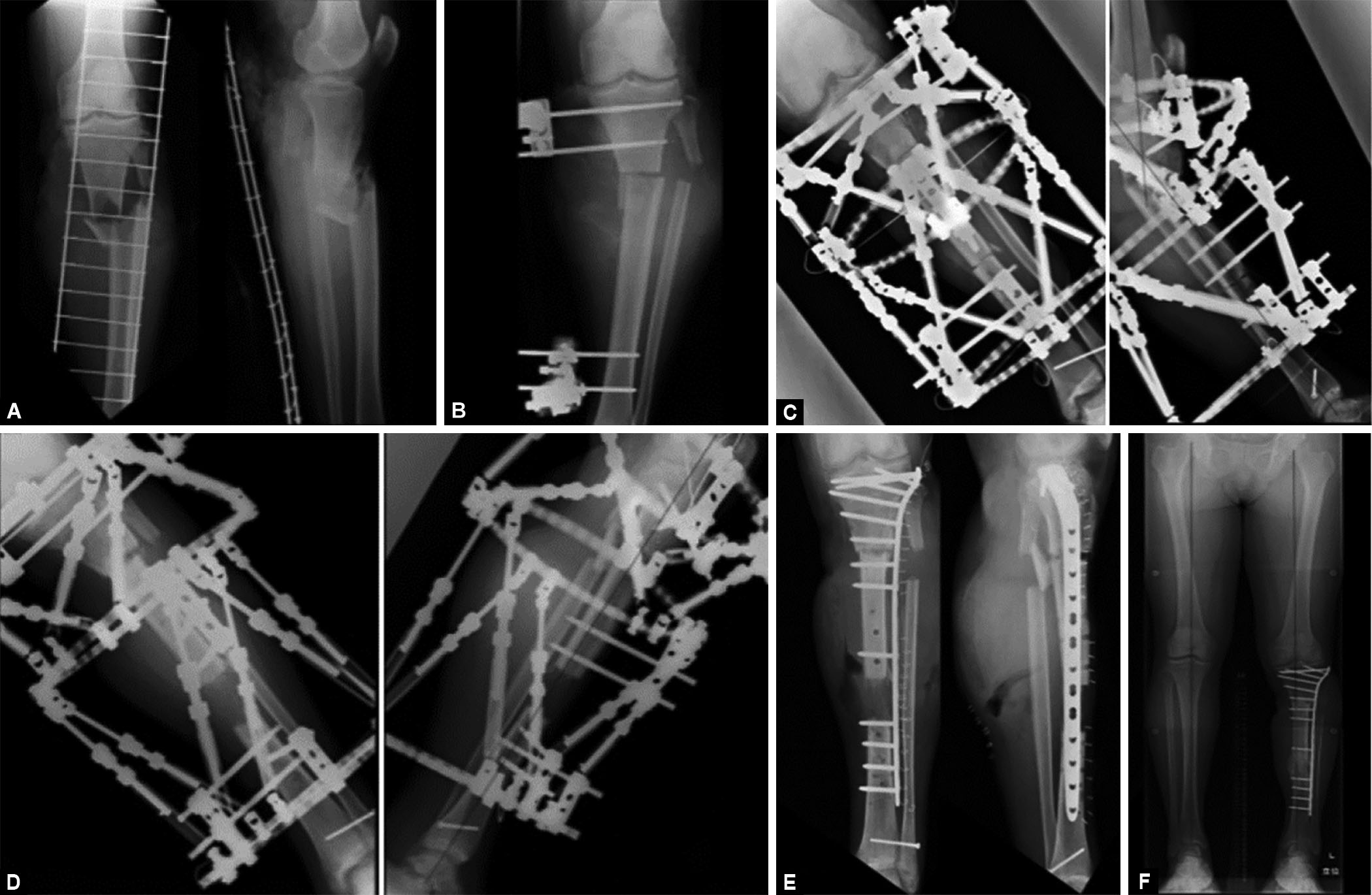
Figs 2A to F: Progress of X-ray – Case 1. (A) He was injured proximal tibial open fracture; (B) The primary doctor performed shortening the tibia 3 cm, and application monopolar EF; (C) Deforming the leg through the fracture site into 25° apex posterior, and performed osteotomy distal part of tibia for lengthening; (D) After deformity correction and lengthening; (E) Performing plate conversion; (F) Last follow-up X-ray. Alignment is good and leg length is equal

Figs 3A to C: Progress of wound – Case 1. (A) First consultation; (B) Confirming the soft tissue can be closed by deformation; (C) Last follow-up
Case 2
A 55-year-old male had fallen and suffered a closed fracture at the distal tibia and fibula (Fig. 4A). The first surgeon had performed an open reduction and internal fixation with a plate on the lateral malleolus and anteromedial tibia 9 days after the injury (Fig. 4B). This wound became infected. Despite repeated debridement, the infection had not improved; the plates were removed at 2 months postoperatively (Fig. 4C). The patient was referred to our institution for treatment. At the first consultation, the tibia, tibialis anterior, and peroneal tendons were found exposed (Fig. 5A). One wound was anterior 10 cm × 3 cm and the other wound was lateral 9 cm × 3 cm. Negative-pressure wound therapy was commenced initially in an attempt to reduce the soft tissue defects. At definitive surgery, the medial malleolus that was un-united was removed. The TSF was applied and, to achieve primary closure of the wound (Fig. 5B), the foot was moved 4° valgus, 31° dorsiflexion, and 10-mm anterior translation (Fig. 4D). The wound was then closed. A proximal tibial lengthening was planned to occur concurrently with a correction of the deformity distally after a wait-period of 2 weeks (Fig. 4E). Three months after the operation, good alignment and 50-mm lengthening had been achieved. The wound was healed completely at 3 months (Fig. 5C). Four months after the first operation, conversion of fixation to plate and screws was performed. However, the distal docking site showed a non-union. Eight months after the first operation, we performed arthrodesis at the subtalar and talocrural joint using a retrograde nail and iliac crest bone graft (Figs 4F and G). At review of 4 years following the first operation, the alignment was assessed to be good, there was no leg length discrepancy (Fig. 4H), and the patient walked without a limp. The bone and functional scores were excellent according to the modified criteria of Paley et al.

Figs 4A to H: Previous hospital and our hospital treatment – Case 2. (A) He suffered from closed pilon fracture; (B) Previous hospital performed plate osteosynthesis; (C) The plate was removed due to infection 2 months after the first operation; (D) TSF application, removed distal non-union malleolus and osteotomy at proximal part, M–L view; (E) After deformity correction and lengthening; (F and G) Plate conversion and talocrural and subtalar arthrodesis was performed; (H) Leg length is equal

Figs 5A to C: Progress of wound – Case 2. (A) Picture of wounds at first consultation; (B) Picture of first operation. The wounds were closed; (C) After 1.5 months from first operation. The wounds were healing
Case 3
A 21-year-old male had been in a traffic accident and sustained an open tibial shaft fracture (Fig. 6A). A previous surgeon had placed a cement spacer after debridement and applied an EF (Fig. 6B). One month after the injury, he was referred to our institution for treatment of bone and soft tissue defects. A 3 cm × 5 cm soft tissue defect exposing the cement spacer was found distal to the lower tibial shaft (Fig. 7A). Pseudomonas aeruginosa was detected in a wound culture. At surgery, devitalised and contaminated soft tissue was excised, and the cement spacer removed (Fig. 7B). We were able to confirm the soft tissue wound edges could be approximated by intentional shortening and deformation (Fig. 7C) of 10° of varus and 4° of apex–posterior angulation (Fig. 6C). Bone lengthening was performed proximally through an osteotomy and deformation was corrected distally after a 2-week waiting period. The wound was healed completely 1.5 months after the operation (Fig. 7D). Restoration of alignment took approximately 4 months because of the large amount of shortening (Fig. 6D). The leg lengths became equal at 5 months after a 7-cm lengthening (Fig. 6E). As this case was not suitable for plate conversion, iliac bone grafting at the docking site was performed. One year after the operation, bone union was achieved, and the hardware was removed. Two years following the first operation, this patient is able to run, climb stairs, and work as a caregiver (Figs 6F and 7E). The bone and functional outcomes were excellent according to the modified criteria of Paley et al.
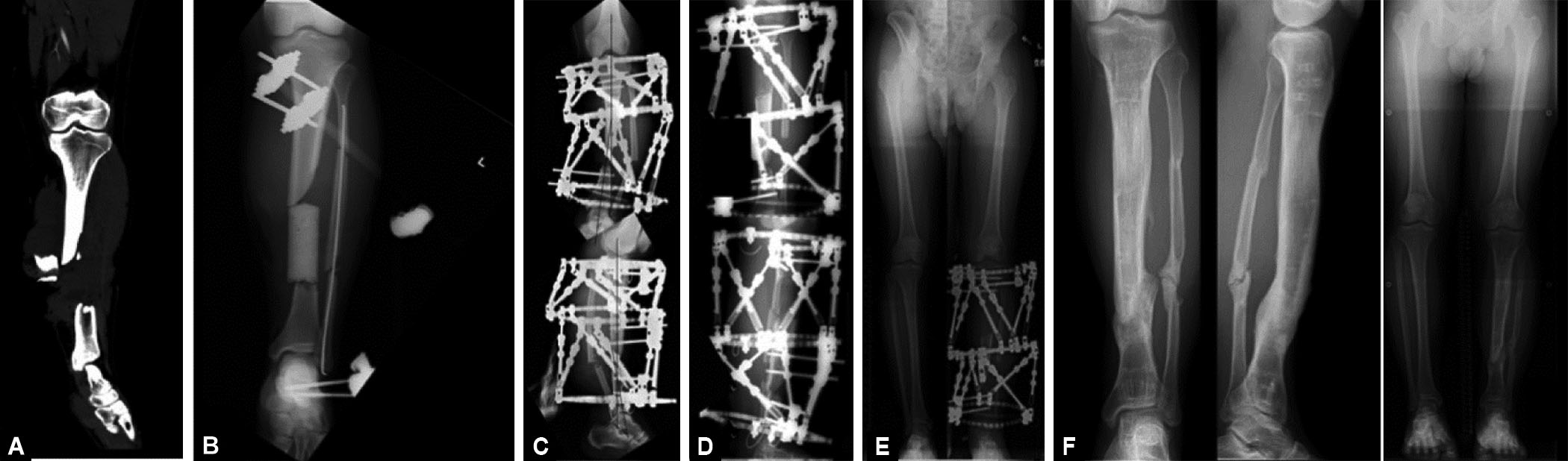
Figs 6A to F: Progress of X-ray – Case 3. (A) He was injured by an open tibial shaft fracture. The third fragment was out of the wound; (B) The previous doctor urgently placed a cement spacer and application EF; (C) Ten degrees of varus and four degrees of posterior apex deformation were performed. Bone lengthening was performed proximally, and deformation was corrected distally; (D) Alignment correction took about 4 months due to the large amount of shortening; (E) Leg length become equal 5 months after operation; (F) Bone union was obtained, alignment is good

Figs 7A to E: Progress of clinical photos – Case 3. (A) A soft tissue defect of 3 cm × 5 cm was found distal to the lower shaft of tibia, exposing the cement spacer; (B) Dirty soft tissue was excised, the cement spacer was removed; (C) It was confirmed that the soft tissue could be covered by shortening and varus deformation; (D) The wound was closed 1.5 months after the operation; (E) At the final follow-up
DISCUSSION
The method of intentional shortening and deformity creation enabled simultaneous treatment of soft tissue and bone defects. Generally, an infected non-union (bone defect) is treated by removing all infected bone and then filling the void with normal bone.4 Vascularised fibula, bone transport, and the Masquelet technique are used for bone reconstruction. Treatment is more difficult when soft tissue defects are present in addition to bone defects.1 Soft tissue reconstruction can be treated with a vascularized fibular and flap but these options have disadvantages. First, a vascularized fibula is associated with complications at both the donor and recipient sites. The recipient site needs to hypertrophy13 and there are risks of stress fractures of the grafted fibula.14 The donor site has a risk of ankle instability15 and valgus deformity.16 Second, bone transport has a high probability of needing bone grafting at the docking site. Therefore, surgery must be performed multiple times. Third, a vascularised free flap depends on the recipient site condition and success is highly dependent on the operator’s skill. Use of a flap always carries the risk of flap loss.17–19
This method can treat bone and soft tissue defects simultaneously by applying deformation and shortening and does not require a flap. This method can also be used to treat patients in whom a flap is not possible. Case 1 in the present study involved a patient with vascular injuries. High rates of flap failure have reportedly been correlated with decreasing numbers of patent vessels in the leg.20 Therefore, a good indication for intentional deformation and shortening is for patients in whom a flap cannot be used because of the vascular status.
The method proposed for simultaneous treatment of bone and soft tissue defects has some disadvantages. First, this method must be used with a ring EF, which is a heavy burden for patients. Second, this method takes a long time. Our Cases 1 and 2, but not Case 3, underwent plate conversion. Therefore, the EF period was much longer for the patient in Case 3 than for the other two patients (Table 1).
This method was reported by Nho et al.8 and was used to treat parts of some open fractures. Subsequently, Gulsen and Ozkan21 used this method to treat infected non-unions (bone defects) in two parts. An unusual aspect of our cases was that Cases 2 and 3 underwent bone transport; there have been no similar reports to date. Moreover, the method we adopted involved the use of three rings of the TSF with the deformity correction performed only at the sites with soft tissue damage and the bone lengthened at the healthy sites. Avoiding bone lengthening at the site of soft tissue injuries can lead to good bone maturation in an area of normal blood flow. The Ilizarov method22,23 is based on “distraction histogenesis” which is a local biological regenerative process. Distraction osteogenesis induces an increased blood flow in the bone lengthening part24,25 which augments the healing of both bone and soft tissue. Treatment at two sites simultaneously also adds to this improved lower-limb blood flow. This is a useful method when the bone defect and soft tissue defect occur simultaneously.26
The important points to note regarding this method are the amount of shortening and deformation. It has been said that 3 cm is the limit for an acute shortening of the tibia.27 Others have reported that middle and distal tibial defects can routinely accommodate up to 6 cm of acute shortening.28,29 In our case series, the maximum shortening was 4 cm. If it is found that the amount of shortening is insufficient to create bone-to-bone contact, this can be adjusted by performing bone transport. Caution is required because neurovascular bundles undergo kinking and normal function may not be possible. It is important to monitor the foot with a pulse oximeter when shortening and producing the deformation.
This case series is small because the conditions for which this method is indicated are very limited. Vascular injuries, such as Gustilo 3C, are rare.30 Further evaluation of cases using such a method is needed and the scarcity of numbers may be overcome by collecting data from multiple centres.
CONCLUSION
This study has showed that intractable cases involving patients with massive bone and soft tissue defects can benefit from reconstruction using intentional deformation and shortening.
ORCID
Hidenori Matsubara https://orcid.org/0000-0002-8394-1003
REFERENCES
1. Grubor P, Milicevic S, Grubor M, et al. Treatment of bone defects in war wounds: retrospective study. Med Arch 2015;69(4):260–264. DOI: 10.5455/medarh.2015.69.260-264.
2. El-Gammal TA, Shiha AE, El-Deen MA, et al. Management of traumatic tibial defects using free vascularized fibula or Ilizarov bone transport: A comparative study. Microsurgery 2008;28(5):339–346. DOI: 10.1002/micr.20501.
3. Arnež ZM, Papa G, Ramella V, et al. Limb and flap salvage in Gustilo IIIC injuries treated by vascular repair and emergency free flap transfer. J Reconstr Microsurg 2017;33(S 01):S03–S07. DOI: 10.1055/s-0037-1603737.
4. Masquelet AC, Fitoussi F, Begue T, et al. Reconstruction of the long bones by the induced membrane and spongy autograft. Ann Chir Plast Esthet 2000;45(3):346–353. PMID: 10929461.
5. Masquelet AC, Begue T. The concept of induced membrane for reconstruction of long bone defects. Orthop Clin North Am 2010;41(1):27–37. DOI: 10.1016/j.ocl.2009.07.011.
6. Ronga M, Ferraro S, Fagetti A, et al. Masquelet technique for the treatment of a severe acute tibial bone loss. Injury 2014;45(Suppl. 6):S111–S115. DOI: 10.1016/j.injury.2014.10.033.
7. Olesen UK, Eckardt H, Bosemark P, et al. The Masquelet technique of induced membrane for healing of bone defects. A review of 8 cases. Injury 2015;46(Suppl. 8):S44–S47. DOI: 10.1016/S0020-1383(15)30054-1.
8. Nho SJ, Helfet DL, Rozbruch SR. Temporary intentional leg shortening and deformation to facilitate wound closure using the Ilizarov/Taylor Spatial Frame. J Orthop Trauma 2006;20(6):419–424. DOI: 10.1097/00005131-200607000-00010.
9. Frazer JE. The skull: General account. In: Breathnaach AS, editor. Anatomy of the human skeleton. 6th ed. London: J and A Churchill Ltd., 1965, Vol. 2, pp. 161–181.
10. Paley D. History and science behind the six-axis correction external fixation devices in orthopaedic surgery. Oper Tech Orthop 2011;21(2):125–128. DOI: 10.1053/j.oto.2011.01.011.
11. Gustilo RB, Anderson JT. Prevention of infection in the treatment of one thousand and twenty-five open fractures of long bones: Retrospective and prospective analyses. J Bone Joint Surg Am 1976;58(4):453–458. PMID: 773941.
12. Johnson EN, Burns TC, Hayda RA, et al. Infectious complications of open type III tibial fractures among combat casualties. Clin Infect Dis 2007;45(4):409–415. DOI: 10.1086/520029.
13. Shi LL, Garg R, Jawa A, et al. Bony hypertrophy in vascularized fibular grafts. Hand 2022;17(1):106–113. DOI: 10.1177/1558944719895784.
14. Gao–Hong R, Run–Guang L, Gui–Yong J, et al. A solution to the vessel shortage during free vascularized fibular grafting for reconstructing infected bone defects of the femur: Bridging with vein transplantation. Injury 2017;48(2):486–494. DOI: 10.1016/j.injury.2016.10.027.
15. Pacelli LL, Gillard J, Mcloughlin SW, et al. A biomechanical analysis of donor-site ankle instability following free fibular graft harvest. J Bone Joint Surg 2003;85(4):597–603. DOI: 10.2106/00004623-200304000-00002.
16. van der Veen FJ, Strackee SD, Besselaar PP. Progressive valgus deformity of the donor-site ankle after extraperiosteal harvesting the fibular shaft in children. Treatment with osteotomy and synostosis at one session. J Orthop 2014;12(Suppl. 1):S94–S100.
17. Ricci JA, Abdou SA, Stranix JT, et al. Reconstruction of Gustilo Type 3C injuries of the lower extremity. Plast Reconsrt Surg 2019;144(4):982–987. DOI: 10.1097/PRS.0000000000006063.
18. Stoddart MT, Al-Hourani K, Fowler T, et al. Plate-assisted intramedullary nailing of Gustilo type IIIB open tibial diaphyseal fractures: does adjunctive plate retention affect complication rate? J Orthop Trauma 2020;34(7):363–369. DOI: 10.1097/BOT.0000000000001738.
19. Houdek MT, Wagner ER, Watts CD, et al. Free composite serratus anterior-latissimus-rib flaps for acute one-stage reconstruction of Gustilo IIIB tibia fractures. Am J Orthop 2018;47(6). DOI: 10.12788/ajo.2018.0047.
20. Ricci JA, Stranix JT, Lee ZH, et al. Comparing reconstructive outcomes in patients with Gustilo type 3B fractures and concomitant arterial injuries. Plast Reconstr Surg 2019;143(5):1522–1529. DOI: 10.1097/PRS.0000000000005552.
21. Gulsen M, Ozkan C. Angular shortening and delayed gradual distraction for the treatment of asymmetrical bone and soft tissue defects of tibia: A case series. J Trauma 2009;66(5):E61–E66. DOI: 10.1097/TA.0b013e318031cca8.
22. Ilizarov GA. The tension–stress effect on the genesis and growth of tissues. Part I. The influence of stability of fixation and soft-tissue preservation. Clin Orthop Relat Res 1989;(238):249–281. PMID: 2910611.
23. May JW, Jupiter JB, Weiland AJ, et al. Clinical classification of post-traumatic tibial osteomyelitis. J Bone Joint Surg Am 1989;71(9):1422–1428. PMID: 2677014.
24. Sakurakichi K, Tsuchiya H, Watanabe K, et al. Distraction osteogenesis of a fresh fracture site using an external fixator. J Orthop Sci 2006;11(4):390–393. DOI: 10.1007/s00776-006-1022-0.
25. Minematsu K, Tsuchiya H, Taki J, et al. Blood flow measurement during distraction osteogenesis. Clin Orthop Relat Res 1998;(347):229–235. PMID: 9520895.
26. Lahoti O, Findlay I, Shetty S, et al. Intentional deformation and closure of soft tissue defect in open tibial fractures with a Taylor spatial frame: A simple technique. J Orthop Trauma 2013;27(8):451–456. DOI: 10.1097/BOT.0b013e318284727a.
27. Pierrie SN, Hsu JR. Shortening and angulation strategies to address composite bone and soft tissue defects. J Orthop Trauma 2017;31(Suppl. 5):S32–S35. DOI: 10.1097/BOT.0000000000000976.
28. El-Rosasy MA. Acute shortening and re-lengthening in the management of bone and soft-tissue loss in complicated fractures of the tibia. J Bone Joint Surg Br 2007;89(1):80–88. DOI: 10.1302/0301-620X.89B1.17595.
29. Sen C, Kocaoglu M, Eralp L, et al. Bifocal compression–distraction in the acute treatment of grade III open tibia fractures with bone and soft-tissue loss: A report of 24 cases. J Ortho Trauma 2004;18(3):150–157. DOI: 10.1097/00005131-200403000-00005.
30. Chummun S, Wigglesworth TA, Young K, et al. Does vascular injury affect the outcome of open tibial fractures? Plast Reconstr Surg 2013;131(2):303–309. DOI: 10.1097/PRS.0b013e3182789bcf.
________________________
© Jaypee Brothers Medical Publishers. 2022 Open Access This article is distributed under the terms of the Creative Commons Attribution-Non Commercial-share alike license (https://creativecommons.org/licenses/by-nc-sa/4.0/) which permits unrestricted distribution, and non-commercial reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. If you remix, transform, or build upon the material, you must distribute your contributions under the same license as original. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.